Insmed’s 80X80 CHEST Exhibit
In August 2025, the FDA approved BRINSUPRI as the first and only treatment for non-cystic fibrosis bronchiectasis, a serious, chronic lung disease.
“The FDA approval of the first-ever treatment for non-cystic fibrosis bronchiectasis is a historic milestone for patients and for Insmed,” said Martina Flammer, M.D., MBA, Chief Medical Officer of Insmed. “By keeping patients at the center of everything we do, we have once again delivered a first-in-class medicine for a disease with no prior approved treatments. This is an incredible achievement in medicine. We’re deeply grateful to the patients, providers, and advocates who made this possible – this is just the beginning of what we can accomplish together for this community.”
CHEST 2025 in Chicago was the marquee professional moment, the first major respiratory congress since approval, and Insmed presented significant scientific data. The breadth of data presentations signaled not only efficacy but also a long-term commitment to building the evidence base around non-cystic fibrosis bronchiectasis (NCFB) as a category.
The Experience
Because of an increased incidence of NCFB, it is no longer considered a rare disease. FDA-approved BRIINSUPRI and Insmed’s launch at CHEST2025 were welcome additions to the available therapies that improve breathing. The exhibit was designed by Area 23 and fabricated by Access, featuring a hanging sphere visible across the floor. A smaller floor-mounted replica of the sphere included a touchscreen interface that displayed the mechanism of action. Patient stories bolstered the message that help is now available for NCFB.
The exhibit presented a calm park-like environment with plenty of comfortable seating. There was a section of the exhibit dedicated to Arikayce, Insmed’s prescription, FDA-approved inhaled antibiotic used to treat refractory mycobacterium avium complex (MAC) lung disease in adults with limited or no alternative treatment options.
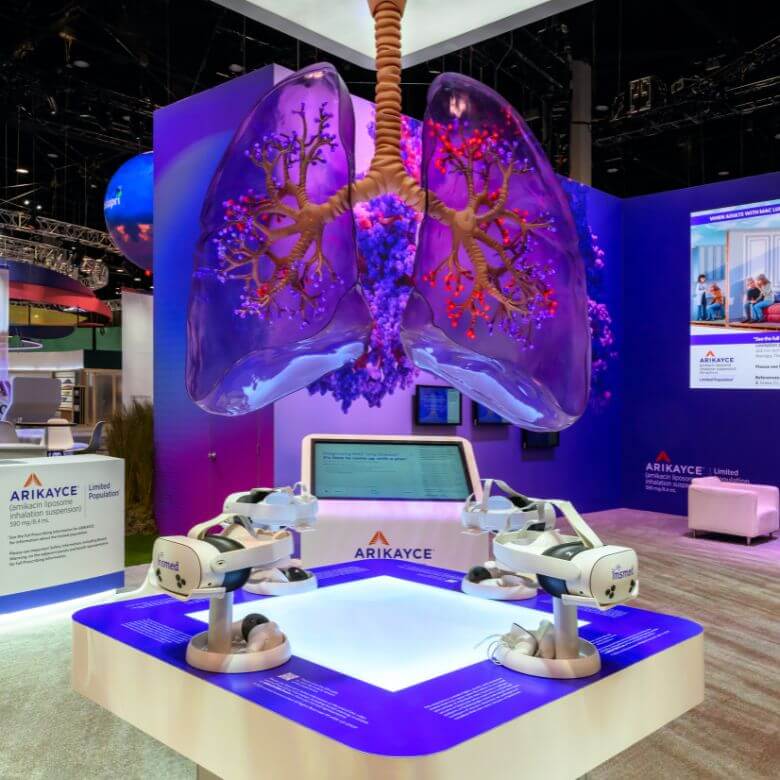
Arikayce featured suspended transparent lungs and the slogan “Break the Pattern!” to attract attendees.

An unbranded medical information area in the exhibit showcased a hand holding a magnifying glass, symbolizing Insmed’s research in finding new disease treatments.

Overhead signs directed HCPs to the Insmed exhibit and aided their navigation.

























